|
You wrap the head of the match and about half way up the stick in several layers of foil, being sure to seal the part around the head well. I remember years ago, I couldn't have been more than ten, making rockets out of matches and tin (aluminum) foil. Strike-on-box matches contain phosphorus only in the striking surface, not in the match itself. These are strike-anywhere matches which contain a small amount of red phosphorus in the head. Caution! Pyrophoric! May spontaneously ignite with friction! Wear appropriate protective gear when handling this substance! Never expose to heat or rough handling! Probably the second most dangerous element I have after cesium. Source: Tryggvi Emilsson and Timothy BrumleveĬontributor: Tryggvi Emilsson and Timothy Brumleveīoy, if these were chemicals the warnings on them would be a mile wide. Funny how the bonding structure can make such a difference.Ĭlick the source link for an interesting story about where this sample came from. It's the form that glows in the dark because of the spontaneous reaction with air on its surface. Not so white phosphorus: That has to be kept under water, or preferably not kept at all. 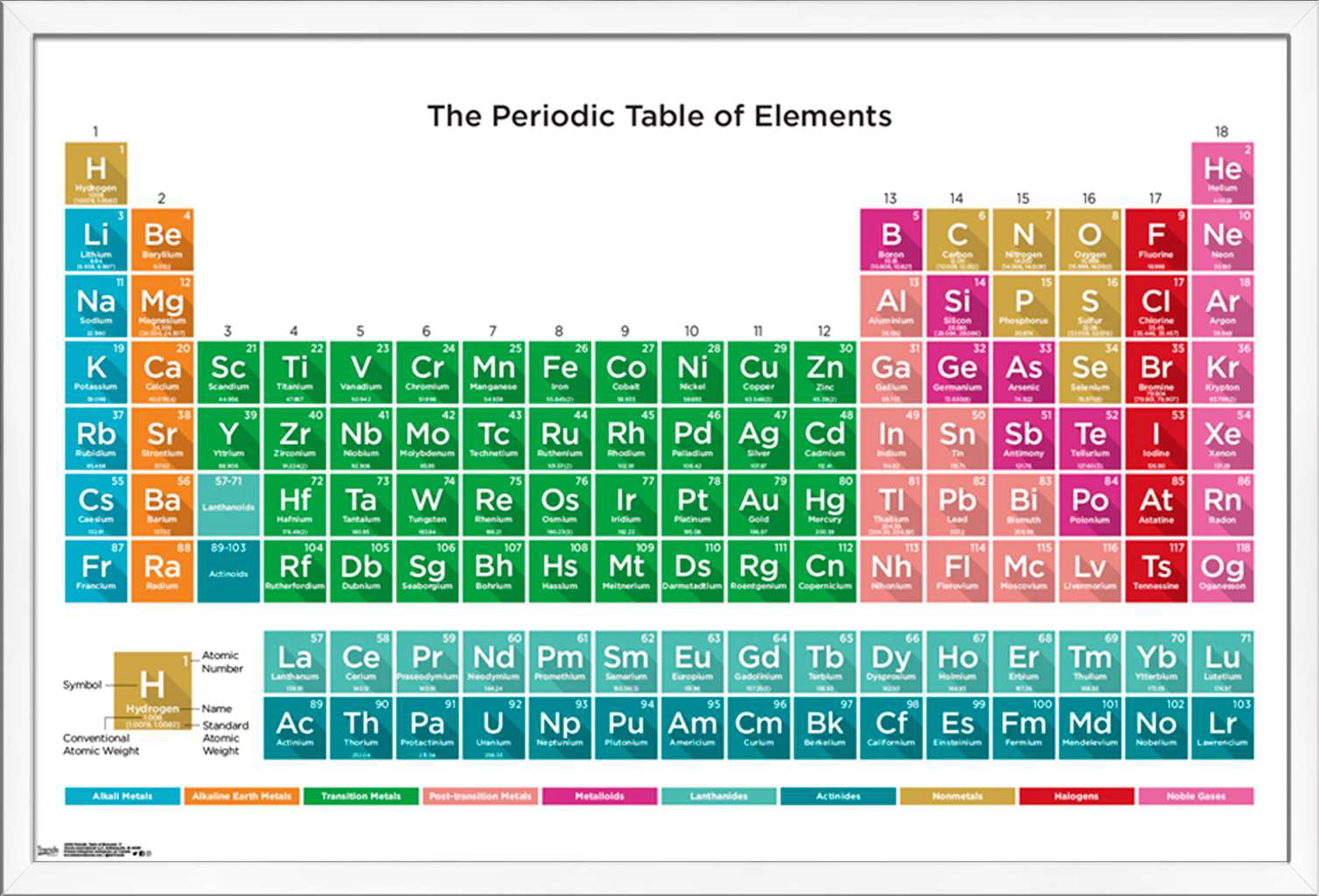
Red phosphorus is reasonably safe, and not likely to spontaneously explode or anything. Phosphorus comes in several allotropic forms. Here is the company's version (there is some variation between sets, so the pictures sometimes show different variations of the samples): Or you can see both side-by-side with bigger pictures in numerical order. You can see photographs of all the samples displayed in a periodic table format: my pictures or their pictures. 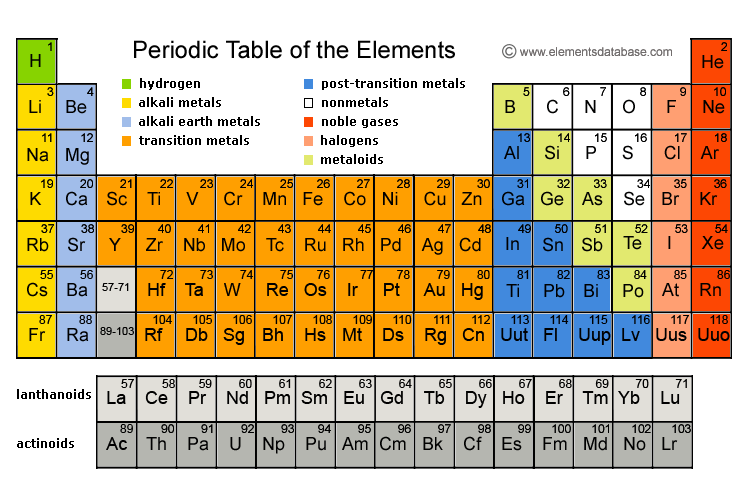
I have two photographs of each sample from the set: One taken by me and one from the company. To learn more about the set you can visit my page about element collecting for a general description or the company's website which includes many photographs and pricing details. Max Whitby, the director of the company, very kindly donated a complete set to the periodic table table. The Red Green and Blue company in England sells a very nice element collection in several versions. To learn more about the set you can visit my page about element collecting for a general description and information about how to buy one, or you can see photographs of all the samples from the set displayed on my website in a periodic table layout or with bigger pictures in numerical order. The samples (except gases) weigh about 0.25 grams each, and the whole set comes in a very nice wooden box with a printed periodic table in the lid. 
At some point their American distributor sold off the remaining stock to a man who is now selling them on eBay. Up until the early 1990's a company in Russia sold a periodic table collection with element samples. Dave found a bunch of surplus elements in their chemical storeroom, and naturally Max Whitby and I graciously offered to take them off his hands. This sample was donated by Dave Roberts of DePauw University, who I got to know while I was installing the beautiful periodic table display that now graces their Julian Science Center. 
This is fairly safe red phosphorus ampouled under argon to keep it dry and clean. Red Phosphorus powder ampouled under argon. The sample photograph includes text exactly as it appears in the poster, which you are encouraged to buy a copy of. I chose this sample to represent its element in my Photographic Periodic Table Poster. Violet phosphorus is a mixture of the red and black allotropes, quite unusual, and it was made by Max Whitby himself using one of the strange recipes of heat, pressure, and catalysts needed to transform phosphorus among its various crystal structures. Phosphorus has all sorts of allotropes (different physical forms of the same element), which come in many colors: Red, black, and white being the only real ones (and white being the most dangerous by far). These were more important to have back when matches were less well developed and more likely to go off by accident. Iron match safe designed to hold matches. Scroll down to see examples of Phosphorus.Ĭlick here to buy a book, photographic periodic table poster, card deck, or 3D print based on the images you see here! This exotic violet form is a mixture of red and black, not a true allotrope itself. Phosphorus occurs in white (extremely dangerous), red (safer and common in matches) and black (rare, most stable) forms. Pictures, stories, and facts about the element Phosphorus in the Periodic Table H
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
